Research
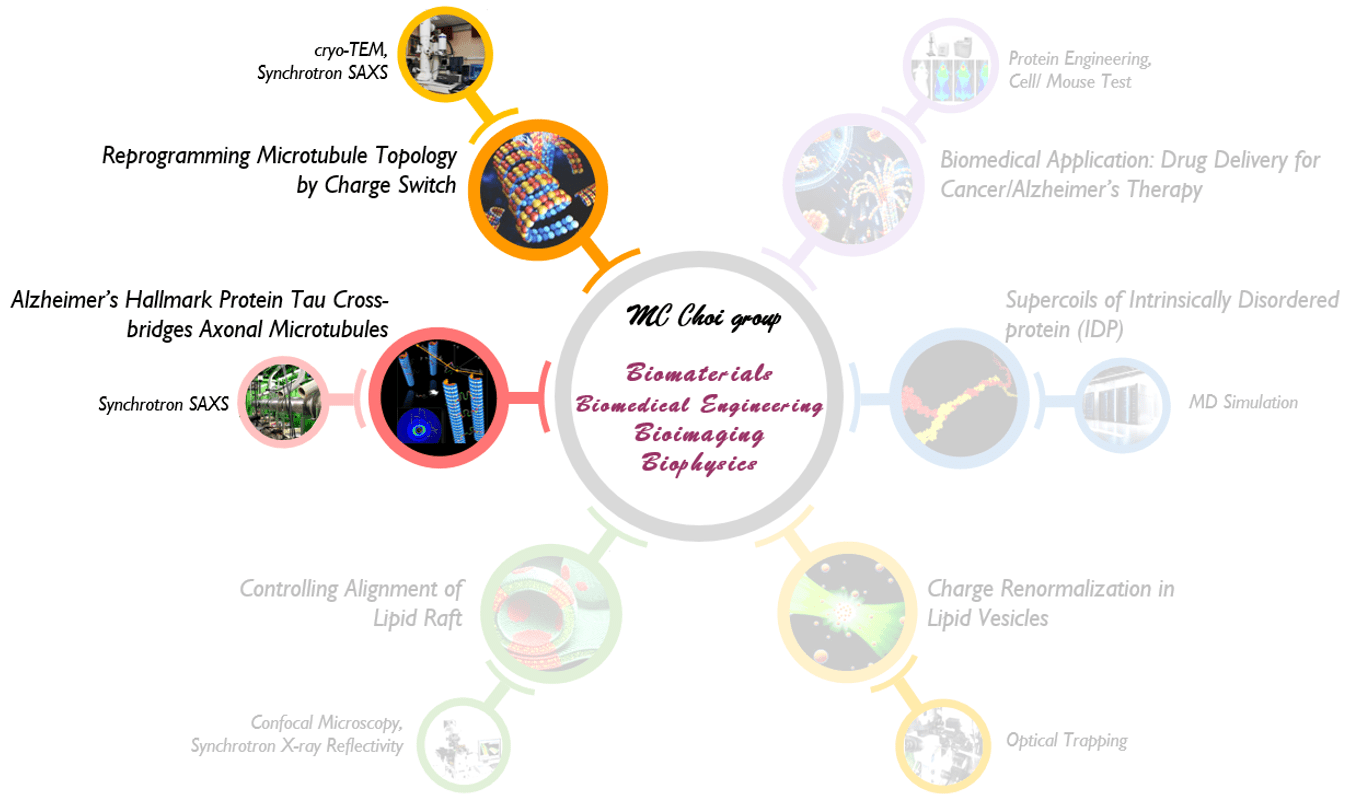
1. Reprogramming Microtubule Topology by Charge Switch; Alzheimer’s Hallmark Protein Tau Cross-bridges Axonal Microtubules
We study on elucidating structures and interactions of microtubules and microtubule-associated-proteins. Microtubules (MTs), hollow 25 nm diameter protein nanotubes, are among the major filamentous elements of the eukaryotic cytoskeleton involved in a range of cellular functions including intracellular trafficking, cell division and the establishment and maintenance of cell shape. A variety of different microtubule-associated-proteins (MAPs) is required for the establishment of neuronal cell polarity, and the maintenance of neuronal axons. Aberrant tau action has long been correlated with numerous neurodegenerative diseases, including Alzheimer’s, Pick’s, supra-nuclear palsy, and fronto-temporal dementia with Parkinsonism linked to chromosome 17 (FTDP-17).
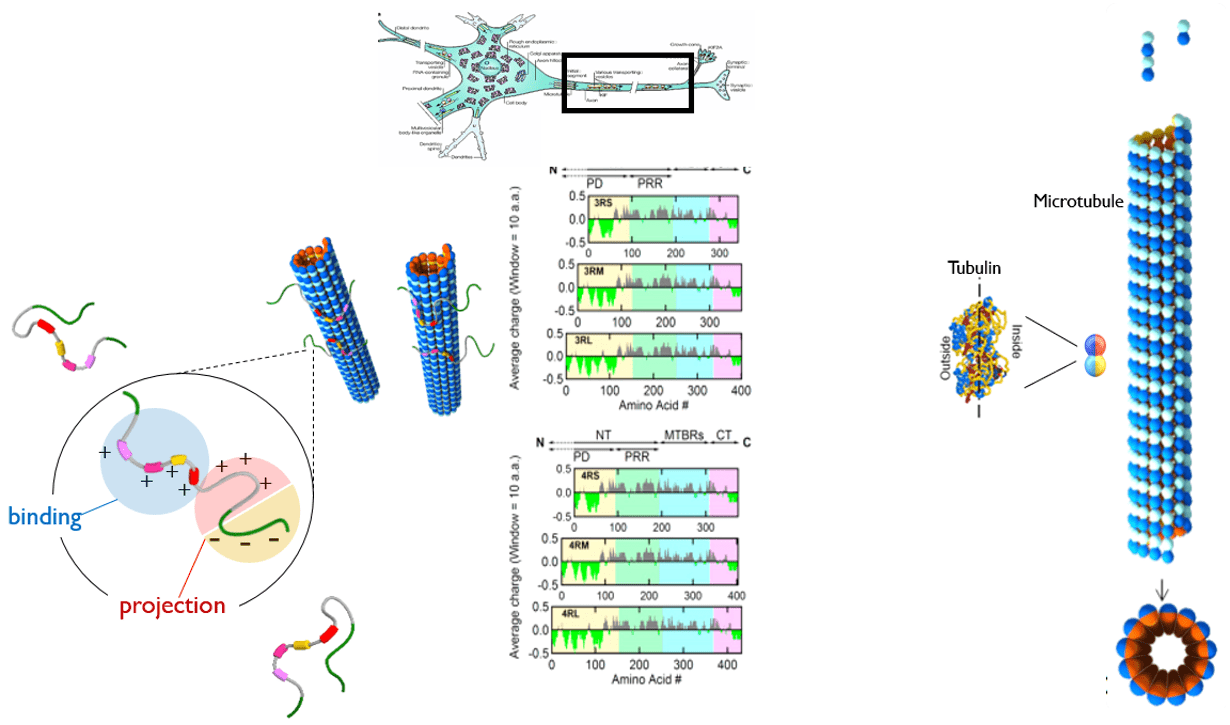
The fundamental understanding of interactions between MTs and MAPs would provide insight for therapeutic applications, and further for controlling assembly structure of biological nanotubes for bioengineering applications.
Nanometer to mesoscopic (many micrometers) scaled structures are solved by combining reciprocal space and real space data resulting from x-ray scattering, differential interference contrast (DIC) and three dimensional confocal microscopy, and electron microscopy (EM) experiments.

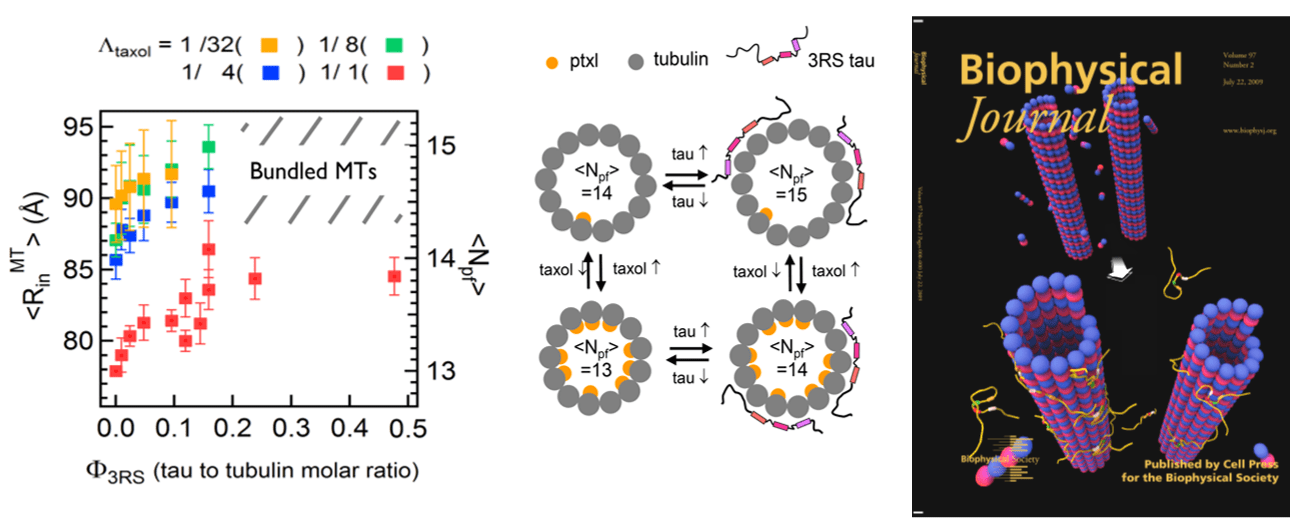
- Alzheimer’s Hallmark Protein Tau Regulates Protofilament Number of Microtubules: MC Choi et al. Biophys J (2009); MC Choi et al. Biochim Biophys Acta (2016)
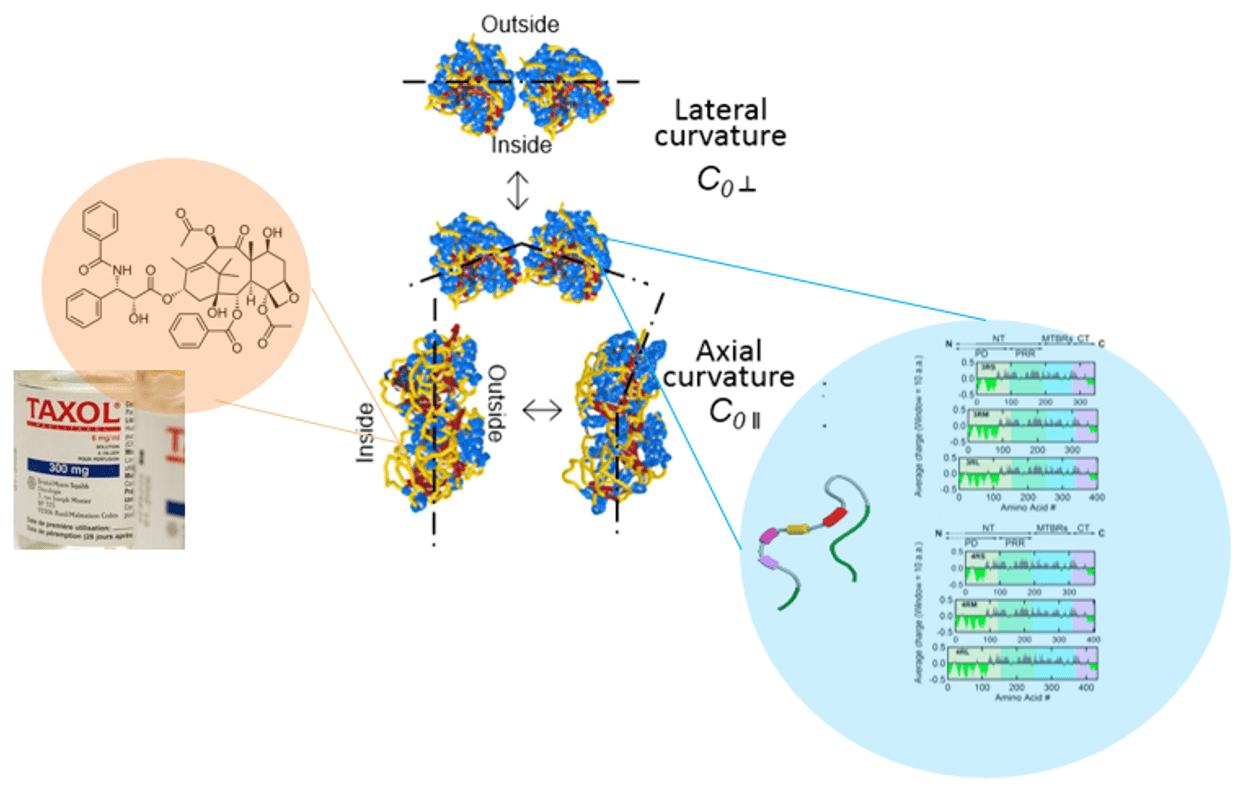
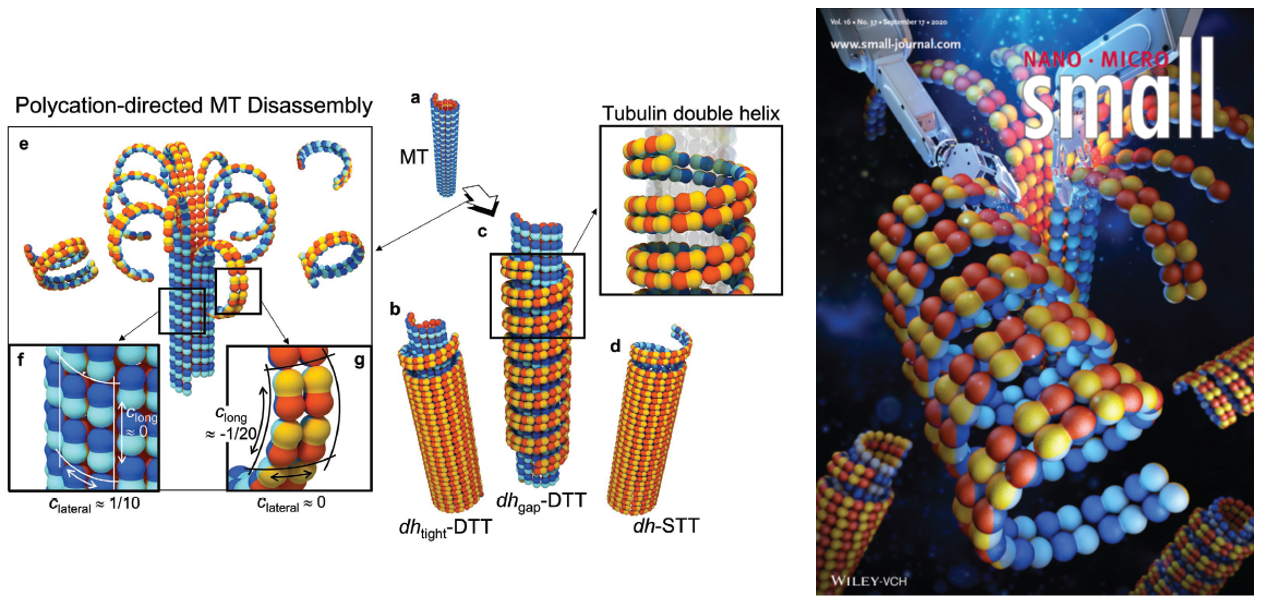
- Tau and Taxol Regulates Structures of Microtubules: MC Choi et al. PNAS (2015); C Safinya et al. Adv. Colloid Interface Sci. (2016); Nat Comm 7 12278 (2016); MC Choi et al. Biochim Biophys Acta (2016)
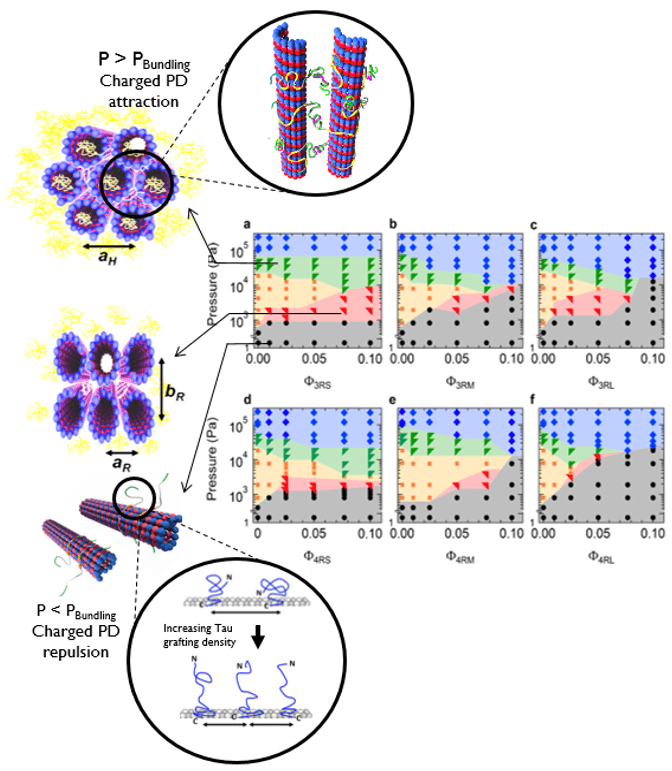
- Reprogramming of Topology of Microtubules by Charge Switch: J Lee et al. Small (2023)
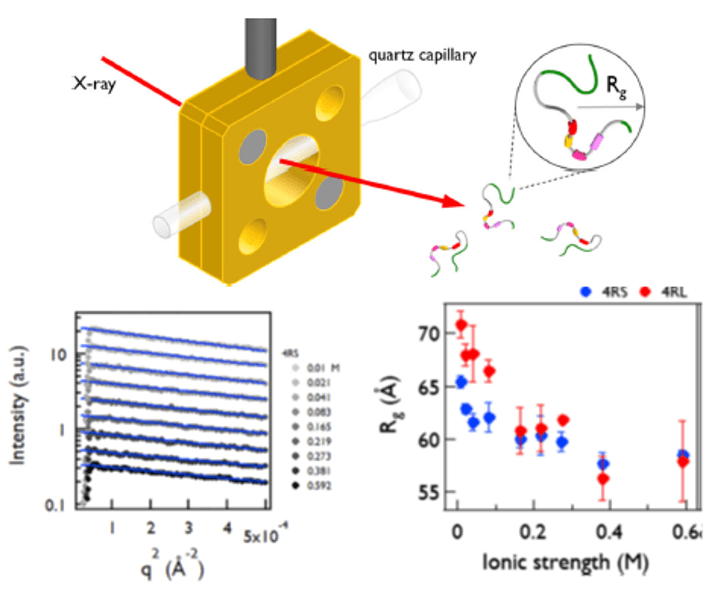
- Intrinsically disordered and polyampholytic nature of Tau: H Cho et al. EPJE (2023)

2. Biomedical Application: Drug Delivery Platform for Cancer/ Alzheimer’s Therapy.
Although microtubule-stabilizing/destabilizing anti-tubulin agents (ATAs) are the most widely used chemotherapeutics in the clinic for treating a wide spectrum of tumors, it has been a challenge to develop appropriate drug-delivery system for such extremely hydrophobic drugs. Furthermore, few drug-delivery carriers capable of delivering more than two ATAs simultaneously are available, despite the fact that these drugs are known to exert synergistic anticancer effects for maximal therapeutic outcome. We develop a delivery platform based on tubulin-derived self-assembled nanostructures that are capable of simultaneously delivering two or more ATAs to cancer cells, a system that could also be generally applied to most ATAs with extremely low water solubility and high systemic toxicities.
We create novel nano-sized architecture “tubulin-based nanotubes (TNTs)” through assembly of tubulin dimers upon interacting with a polycation. Since most ATAs bind tightly to specific sites in tubulin, we hypothesized that TNTs could be used as general delivery carriers for ATAs. Docetaxel (DTX) and Laulimalide (LMD), two microtubule-stabilizing agents that occupy different binding sites in tubulin, were simultaneously loaded onto TNTs. This loading triggered structural changes in TNTs, characterized by shape anisotropy and tubulin layering. In in vitro studies, we showed that TNTs loaded with both drugs were capable of delivering their payloads to cellular tubulins via the endo-lysosomal pathway, resulting in synergistic anticancer and anti-angiogenic effects. This work suggests the new concept of using drugs’ target proteins as a carrier. Our findings further demonstrate that the TNTs developed herein can be used as a delivery platform for microtubule-stabilizing drugs, and potentially for other ATAs, with enhanced therapeutic potency.
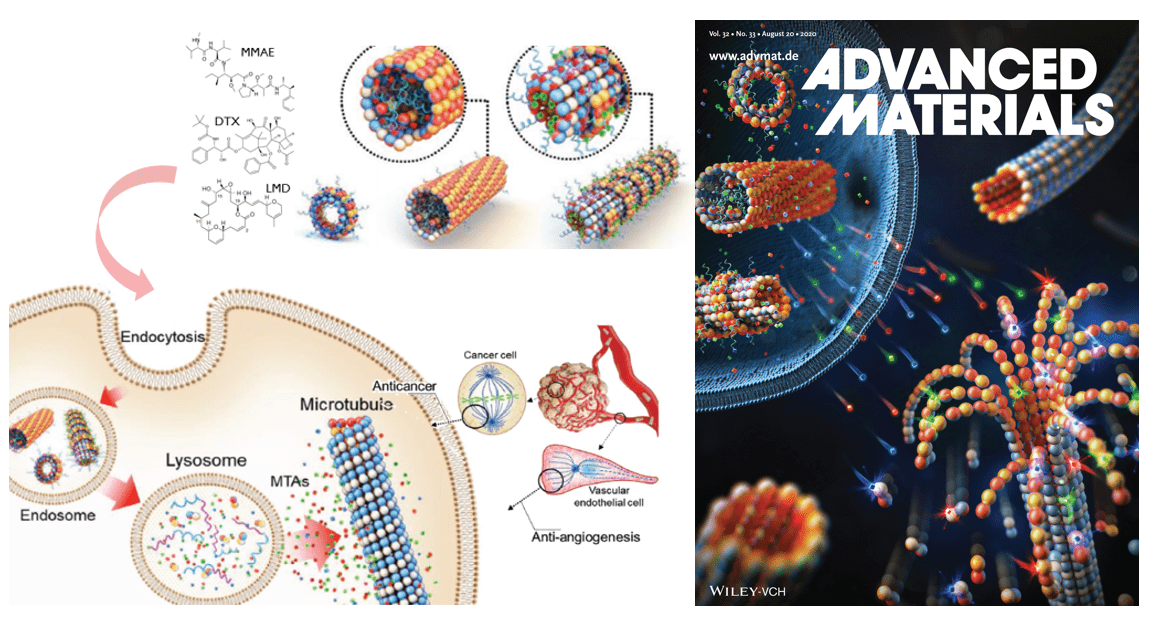
- Using Target protein as Carrier: New Strategy for Anticancer Drug Delivery: J Lee et al. Adv Mat (2020)
3. Controlling Alignment of Lipid Raft.
We study the physical origin of the out-of-plane alignment of lipid rafts, a dynamic nano-domain within the plasma membrane with pivotal roles in intermembrane interactions, including ion channel activation, SNARE-mediated fusion, immune cell pairing, and virus entry. Despite the importance in numerous biological phenomena, single lipid raft in cell membrane is generally small and highly dynamic. Therefore, clustering of lipid rafts with physiologically relevant spatial and temporal scales is required to bring functionally related proteins into close proximity, thus enabling them to interact more effectively. However, the key regulating factor and the molecular driving force governing their out-of-plane alignment across different bilayers has remained elusive compared to extensively studied in-plane growth.
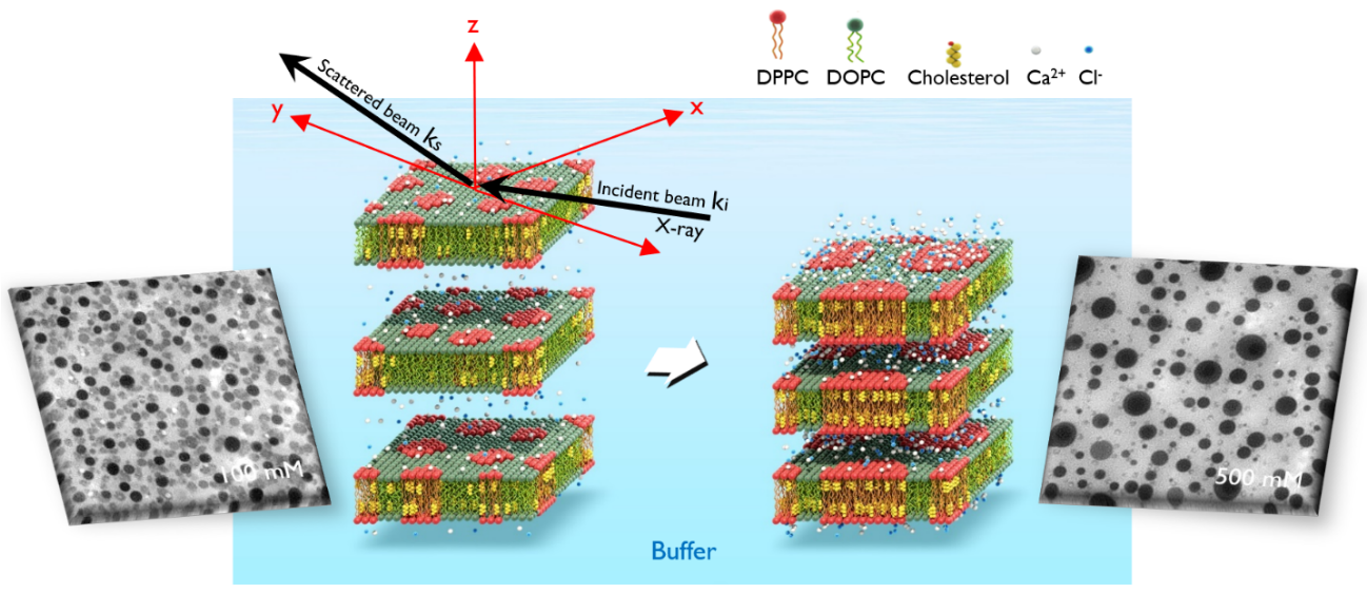
We employed a multi-faceted approach combining fluorescence imaging, synchrotron X-ray reflectivity experiments, and molecular dynamics simulations to investigate the out-of-plane alignment of phase separated lipid multilayers, a model system for studying lipid rafts. They reveal that the alignment of Lo domains is regulated by the intermembrane distance. This domain alignment facilitates in-plane domain growth as well, leading to the formation of stable columnar clusters consisting of large Lo domains. Molecular dynamics simulations reveal a clear preference for the aligned state over the misaligned state when the intermembrane separation is small.

- Proximity for unity: small intermembrane distance as a precursor of lipid rafts alignment: S Lee et al. JACS (2024)
4. Measuring Interactions between Liposomes via Optical Trapping; Charge Renormalization in Lipid Vesicles.
We study the interactions between charged vesicles by using newly developed optical tweezer, called “optical bottle” technique. When a laser beam is applied to the small unilamellar vesicles, the radial forces are exerted on them via optical pressure, thus concentrating vesicles toward the beam center. We measure the second virial coefficient B2 (which is characteristic of the interaction potential) between the vesicles with different surface charge density controlled by a ratio of charged lipids and neutral lipids.

- Probing Inter-Vesicle Force by Optical Trapping: S Park et al. Soft Matter (2014); J Lee et al. To be submitted (2024)